Overview
Lithium batteries are now used in many fields, and in the past, lead-acid batteries, cadmium batteries, and nickel batteries were used in these fields. This article will introduce the development history of lithium-ion batteries and related knowledge of lithium ion battery structure, aimed at readers who are interested in or have purchasing needs for lithium-ion batteries, to help them make wiser choices when purchasing batteries.

1. The development history of lithium-ion batteries
In the past decade, lithium-ion batteries have become the dominant rechargeable battery chemical material in almost all industries. Compared to the previously popular chemical materials (lead-acid batteries, nickel cadmium batteries, and alkaline batteries), lithium-ion batteries are superior in many aspects. Lithium is currently the chemical material with the highest energy density used, and with some additional functions, it can become the safest chemical material. Lithium energy is an active research field, therefore new chemical materials are developed every year.
The concept of lithium-ion batteries was first proposed in the 1970s, when British chemist Stanley Whittingham invented a battery that could charge itself over time. He attempted to use titanium disulfide and lithium metal as electrodes, but this caused the battery to short-circuit and explode.
The safety issues of lithium metal batteries have prompted the development of lithium-ion batteries. Although lithium metal batteries have a higher energy density, lithium-ion batteries are very safe when charging and discharging using specific safety guidelines.
In the 1980s, John Goodenough and Akira Yoshino further experimented to make batteries safer. The development of lithium-ion batteries has thus begun.

In the 1990s, lithium-ion technology began to be favored and rapidly popularized. At that time, Sony produced the first batch of commercial batteries, marking the beginning of commercialization of lithium-ion batteries. At the same time, the portable electronic device market is rapidly growing, requiring a lightweight rechargeable battery to power it. Lithium ion batteries, as a safe and powerful battery, have become the best choice.
In the past decade, lithium-ion batteries have become the dominant rechargeable battery chemical material in almost all industries. Compared to the previously popular chemical materials (lead-acid batteries, nickel cadmium batteries, and alkaline batteries), lithium-ion batteries are superior in many aspects. Lithium is currently the chemical material with the highest energy density used, and with some additional functions, it can become the safest chemical material. Lithium energy is an active research field, therefore new chemical materials are developed every year.
Currently, the top five global lithium ion battery applications companies are:
CATL (China)
LG Chem (South Korea)
BYD (China)
Panasonic (Japan)
Samsung SDI (South Korea)
2. Lithium ion Battery Structure
2.1 What is a lithium-ion battery
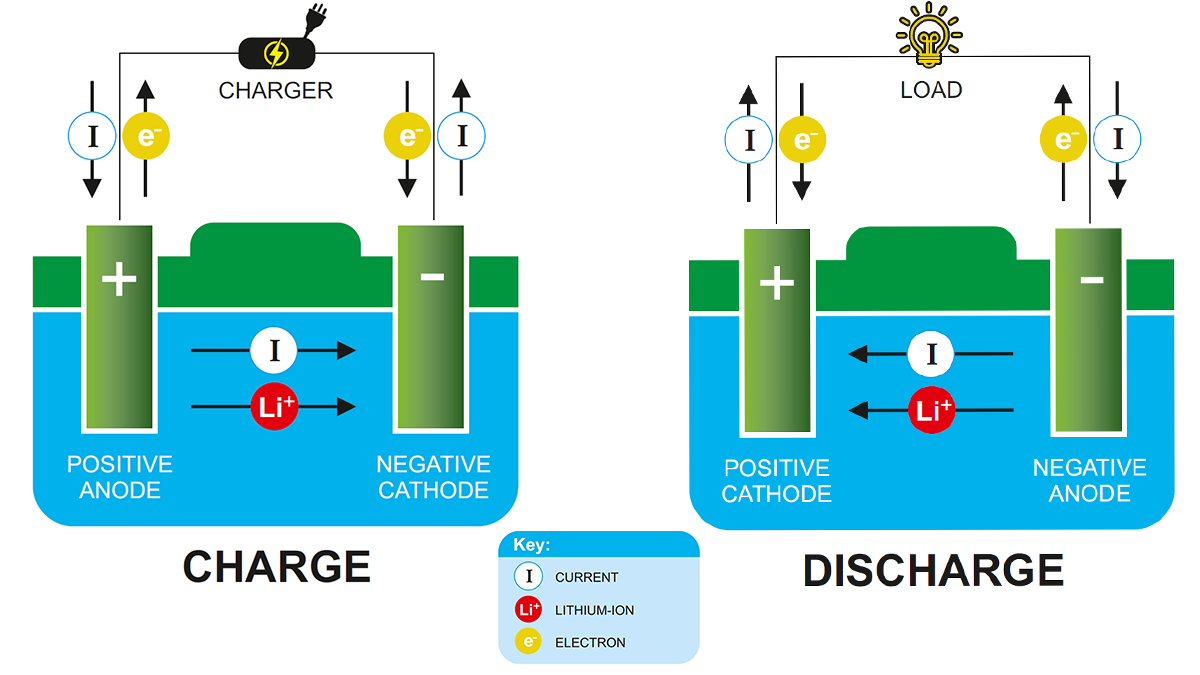
Simply put, a lithium-ion battery refers to a battery with a negative electrode (anode) and a positive electrode (cathode), where lithium ions are transported between the two materials. The working principle of lithium-ion batteries is the same as any other rechargeable battery.
During discharge, lithium ions move from the anode to the cathode and deposit (embed) into the positive electrode composed of lithium and other metals. When charging, this process is the opposite.
Each lithium-ion battery has a voltage range that can operate safely. The range depends on the chemical composition of the electrolyte used in the battery. For example, LFP batteries are 2.5V at 0% state of charge (SOC) and 3.6V at 100% SOC. This is usually considered the safe operating range of LFP batteries, while below the specified range is considered excessive discharge, and exceeding the specified 100% SOC is considered overcharging.
2.2 lithium ion battery structure
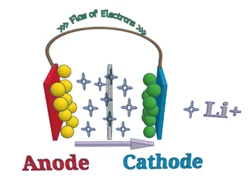
2.2.1 Anode
The anode is the negative electrode in a battery. In lithium-ion batteries, the anode is usually composed of lithium and carbon (usually graphite powder). The purity, particle size, and uniform distribution of anode materials can all affect their capacity and aging rate.
2.2.2 Cathode
The cathode is the positive electrode. This is where different chemicals come into play. The cathode determines the overall lithium energy chemical properties. Like the anode, the collector is combined with the material to facilitate electronic reaction activity. The main difference between them lies in the temperature at which different chemicals react with electrolytes (thermal runaway) and the magnitude of the voltage they produce.
2.2.3 Electrolytes
Electrolytes allow lithium ions to transfer and move between the two plates. Usually, it is composed of different organic carbonates, such as ethylene carbonate and diethyl carbonate. The different mixtures and ratios depend on the application environment of the battery.
For example, for low-temperature applications, the viscosity of the electrolyte solution will be lower than that of the electrolyte solution at room temperature. In lithium batteries, lithium hexafluorophosphate (LiPF6) is the most common lithium salt. It can be said that the most widely used electrolyte in lithium-ion batteries is lithium hexafluorophosphate (LiPF6), whose quality determines the charging and discharging performance, service life, and safety of lithium-ion batteries.
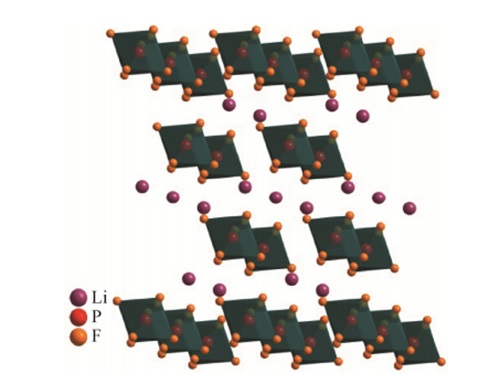
Because LiPF6 has the best overall comprehensive performance, it has excellent environmental friendliness, passivation of the positive electrode current collector to prevent electrode corrosion, and when mixed with water, it produces hydrofluoric acid (HF), which is conducive to the formation of SEI film on the negative electrode.
SEI is a chemical reaction between lithium metal and electrolyte, which forms a solid electrolyte layer on the surface of lithium metal. It plays a role in isolation and protection between lithium metal and electrolyte.
Under normal conditions, battery manufacturers typically charge slowly to form a uniform SEI on the carbon anode.
2.2.4 Diaphragm
The separator of lithium-ion batteries is a porous plastic film that facilitates the prevention of direct contact between the anode and cathode. These thin films are usually 20 microns thick with small pores that allow lithium ions to pass through during charging and discharging processes. Once the battery exceeds the temperature range or experiences a short circuit, this separator will close the pores and prevent lithium ions from passing through, thereby stopping the chemical reaction.
3. The advantages of lithium-ion batteries
3.1 advantages of Lithium ion Battery Structure
1. High rate discharge, stable capacity
2.Fast charging
Lithium ion batteries – charged within 1 hour
Lead acid batteries – over 9 hours
3.Small footprint and strong load-bearing capacity
4.Multiple cycles and long service life
Lithium ion batteries – The cycle life is usually 5000 times, and complete discharge does not affect the cycle life
Lead acid batteries -300 to 500 times, complete discharge will affect their lifespan
5.High energy efficiency
Lithium ion batteries -96% output, 4% heat loss
Lead acid batteries -15% heat loss at 85% output
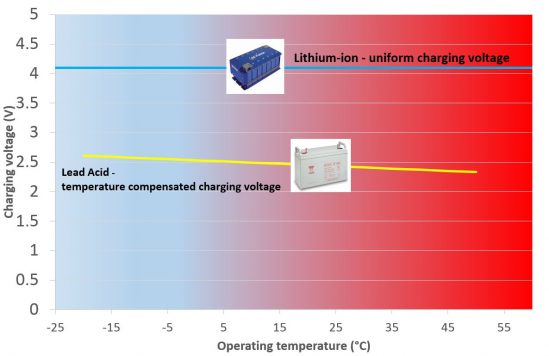
6.Wide range of charging voltage
No voltage compensation required
7.Reduce thermal management costs
Lithium ion batteries – acceptable air circulation
Lead acid battery – requires air conditioning
8.No gas emissions
Lithium ion batteries – operating in sealed containers
Lead acid batteries – require hydrogen ventilation
9.Non toxic, no recycling restrictions
Green new energy, safe and secure to use.
For more comparison between Lithium Battery and Lead Acid Battery, click to view: Lithium Battery vs. Lead Acid Battery for more detailed and specific content.
3.2 The reason for choosing to replace lead-acid batteries with lithium-ion batteries
3.2.1 Improving efficiency
Thanks to advances in BMS and charging technology, lithium-ion battery power supply equipment can help improve efficiency and reduce downtime caused by the need to charge battery powered equipment.
3.2.2 Improving productivity
Operators don’t have to worry about device charging issues, and lithium-ion battery technology allows companies to invest in automation solutions, reducing costs for businesses.
3.2.3 A simpler way to charge and store
Lithium ion batteries can be charged at any time, which means you can charge them at your convenience. Lithium ion batteries also do not require their own charging or storage space, as they do not pose environmental risks like lead-acid batteries.
3.2.4 No maintenance required
Unlike lead-acid batteries, lithium-ion batteries do not require tedious inspections and maintenance methods.
3.2.5 Improving operational safety
Lithium ion batteries improve the operational safety of facilities through various means, and they are also more environmentally friendly due to the lower risk of overheating, explosion, or emission of harmful gases or liquids.
4.1 Balanced charging
Overcharge the battery after a complete charging cycle above normal voltage. This step is necessary to help remove accumulated sulfates and balance the voltage of each battery in lead-acid batteries.
4.2 Battery degradation
The process of reducing the amount of energy that a battery can store. Temperature, charging and discharging voltage, and charging and discharging depth can affect the degree to which battery capacity decreases over time.
4.3 Battery cycle count
If the battery completes one charge and discharge as a cycle, the cumulative number of charges and discharges. The battery cycle consists of 100% discharge and charging.
4.4 Battery life
How long can the battery be used within its lifespan. The lifespan is measured by the number of complete cycles of charging and discharging.
4.5 Working temperature
The acceptable temperature of the surrounding environment in which the battery operates. If the working temperature exceeds the range, the battery may fail.
4.6 UL Listing/Certification
UL listing/certification means that UL has evaluated product samples to ensure they meet specific requirements. This includes testing samples covering the functional safety and use cases of the Lithium ion Battery Structure.
5. Why choose lithium ions- Analysis from the perspective of Lithium ion Battery Structure

5.1 Excellent quality
The high energy density and extensive discharge cycles of lithium-ion batteries are the most important factors, making them indispensable in many devices. And they are also superior to traditional battery chemistry in many other aspects. In addition to the high energy density of batteries, they can also discharge at high power and charge quickly. This gives them greater operational flexibility than lead-acid batteries.
In application scenarios where charging power or time is tight, such as solar photovoltaic systems, continuous operation in partially charged states will not cause harm to lithium-ion batteries.
5.2 Environmentally friendly
The interaction between lithium-ion batteries and the environment is very mild. No harmful gases are emitted during charging, and the heat loss is very low. This means that lithium-ion batteries can be used in enclosed spaces, completely isolated from the surroundings. The recycling and reuse of discarded batteries is also very convenient, as they do not contain toxic substances such as cadmium, mercury, and lead.
5.3 Multiple types of structures
During discharge, the charge moves through the external circuit between the battery electrodes. In order to balance the charge transfer within the battery, positively charged lithium ions move through an internal electrolyte circuit between the positive and negative electrodes. When charging, the process is reversed, and lithium ions return through the electrolyte.
Multiple types of chemicals can be used as cathodes (cathodes) to manufacture electrode materials that carry lithium ions. Electrolyte materials are also a research direction, and the state of materials such as solids and liquids is also a research topic. This is a very active research and development field, which is driving the development of lithium-ion batteries in more and more market applications.
Conclusion
Now that you have understood so much analysis and data, you should have a certain understanding of lithium batteries: their history, advantages, and development direction.
The future of electric products has arrived. The demand for transitioning from traditional energy to new energy cannot be ignored. As more and more industries and enterprises realize the advantages of lithium-ion battery technology, making decisions to shift business focus has become easier.
Contact us, GYCX one-stop service provides the most perfect solution for your needs.
Are you planning to invest in lithium-ion battery technology now?
FAQ
1. Can lithium batteries replace alkaline batteries?
Although lithium batteries use more expensive battery technology, their ability to maintain high voltage means they are an excellent alternative to alkaline batteries.
2.Will lithium batteries leak?
Lithium batteries do not leak, so they are very safe to store.
Lithium can catch fire when in contact with air or water. They are less likely to leak because for liquid electrolytes, exhaust treatment technology is already very mature.
3.At what temperature will lithium-ion batteries explode?
Lithium batteries can explode at 538 degrees Celsius.
If a lithium battery is heated for a long time, it may. Because lithium-ion batteries have a very high energy, when they become hot, they release organic solvents that act on the electrolyte; This heat may cause them to explode.
Short circuits that occur when battery terminals come into contact with metal can also cause explosions.
